Rutherford’s team discovers the nucleus

In 1906 Ernest Rutherford published “Retardation of the α Particle from Radium in passing through matter. Hans Geiger expanded on this work in a communication to the Royal Society with experiments he and Rutherford had done, passing alpha particles through air, foil and gold leaf. More work was published in 1909 by Geiger and Ernest and further greatly expanded work was published in 1910 by Geiger. In 1911–1912 Rutherford went before the Royal Society to explain the experiments and propound the new theory of the atomic nucleus as we now understand it.

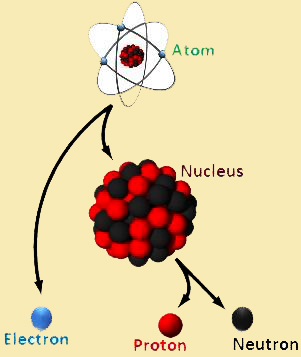
NUCLEAR PHYSICS is the field of physics that studies atomic nuclei and their constituents and interactions. Other forms of nuclear matter are also studied. Nuclear physics should not be confused with atomic physics, which studies the atom as a whole, including its electrons.

Other forms of nuclear matter are also studied. Nuclear physics should not be confused with atomic physics, which studies the atom as a whole, including its electrons.
NUCLEAR PHYSICS nuclear power, nuclear weapons, nuclear medicine and magnetic resonance imaging, industrial and agricultural isotopes, ion implantation in materials engineering, and radiocarbon dating in geology and archaeology. Such applications are studied in the field of nuclear engineering.
Particle physics evolved out of nuclear physics and the two fields are typically taught in close association. Nuclear astrophysics, the application of nuclear physics to astrophysics, is crucial in explaining the inner workings of stars and the origin of the chemical elements.

HERE is the diagram represent the actual meaning of interaction of electrons protons neutrons and the others.
THERE are many parts of the nuclear physics:-
-
Modern nuclear physics
-
Nuclear decay
-
Nuclear fusion
-
Nuclear fission
Firstly we discuss all these four types of nuclear physics.
-
Modern nuclear physics
A heavy nucleus can contain hundreds of nucleons. This means that with some approximation it can be treated as a classical system, rather than a quantum-mechanical one. In the resulting liquid-drop model,the nucleus has an energy which arises partly from surface tension and partly from electrical repulsion of the protons. The liquid-drop model is able to reproduce many features of nuclei, including the general trend of binding energy with respect to mass number, as well as the phenomenon of nuclear fission.
2. Nuclear decay
Eighty elements have at least one stable isotope which is never observed to decay, amounting to a total of about 254 stable isotopes. However, thousands of isotopes have been characterised as unstable. These “radioisotopes” decay over time scales ranging from fractions of a second to trillions of years. Plotted on a chart as a function of atomic and neutron numbers, the binding energy of the nucleus forms what is known as the valley of stability. There are so many nuclear decays like alpha decay,gamma decay.
3.Nuclear fusion
In nuclear fusion, two low mass nuclei come into very close contact with each other, so that the strong force fuses them. It requires a large amount of energy for the strong or nuclear forces to overcome the electrical repulsion between the nuclei in order to fuse them; therefore nuclear fusion can only take place at very high temperatures or high pressures. When nuclei fuse, a very large amount of energy is released and the combined nucleus assumes a lower energy level. The binding energy per nucleon increases with mass number up to nickel-62. Stars like the Sun are powered by the fusion of four protons into a helium nucleus, two positrons, and two neutrinos. The uncontrolled fusion of hydrogen into helium is known as thermonuclear runaway
4.Nuclear fission
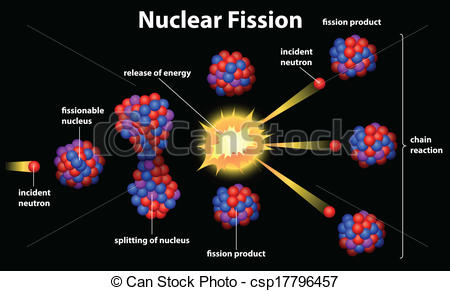
Nuclear fission is the reverse process to fusion. For nuclei heavier than nickel-62 binding energy per nucleon decreases with the mass number. It is therefore possible for energy to be released if a heavy nucleus breaks apart into two lighter ones.
The process of alpha decay is in essence a special type of spontaneous nuclear fission. It is a highly asymmetrical fission because the four particles which make up the alpha particle are especially tightly bound to each other, making production of this nucleus in fission particularly likely.
From certain of the heaviest nuclei whose fission produces free neutrons, and which also easily absorb neutrons to initiate fission, a self-igniting type of neutron-initiated fission can be obtained, in a chain reaction. Chain reactions were known in chemistry before physics, and in fact many familiar processes like fires and chemical explosions are chemical chain reactions. The fission or “nuclear” chain-reaction, using fission-produced neutrons, is the source of energy for nuclear power plants and fission type nuclear bombs, such as those detonated in Hiroshima and Nagasaki, Japan, at the end of World War II. Heavy nuclei such as uranium and thorium may also undergo spontaneous fission, but they are much more likely to undergo decay by alpha decay.

According to the theory, as the Universe cooled after the Big Bang it eventually became possible for common subatomic particles as we know them (neutrons, protons and electrons) to exist. The most common particles created in the Big Bang which are still easily observable to us today were protons and electrons (in equal numbers).
The protons would eventually form hydrogen atoms. Almost all the neutrons created in the Big Bang were absorbed into helium-4 in the first three minutes after the Big Bang, and this helium accounts for most of the helium in the universe today (see Big Bang nucleo synthesis).
Some relatively small quantities of elements beyond helium (lithium, beryllium, and perhaps some boron) were created in the Big Bang, as the protons and neutrons collided with each other, but all of the “heavier elements” (carbon, element number 6, and elements of greater atomic number) that we see today, were created inside stars during a series of fusion stages, such as the proton-proton chain, the CNO cycle and the triple-alpha process. Progressively heavier elements are created during the evolution of a star.
